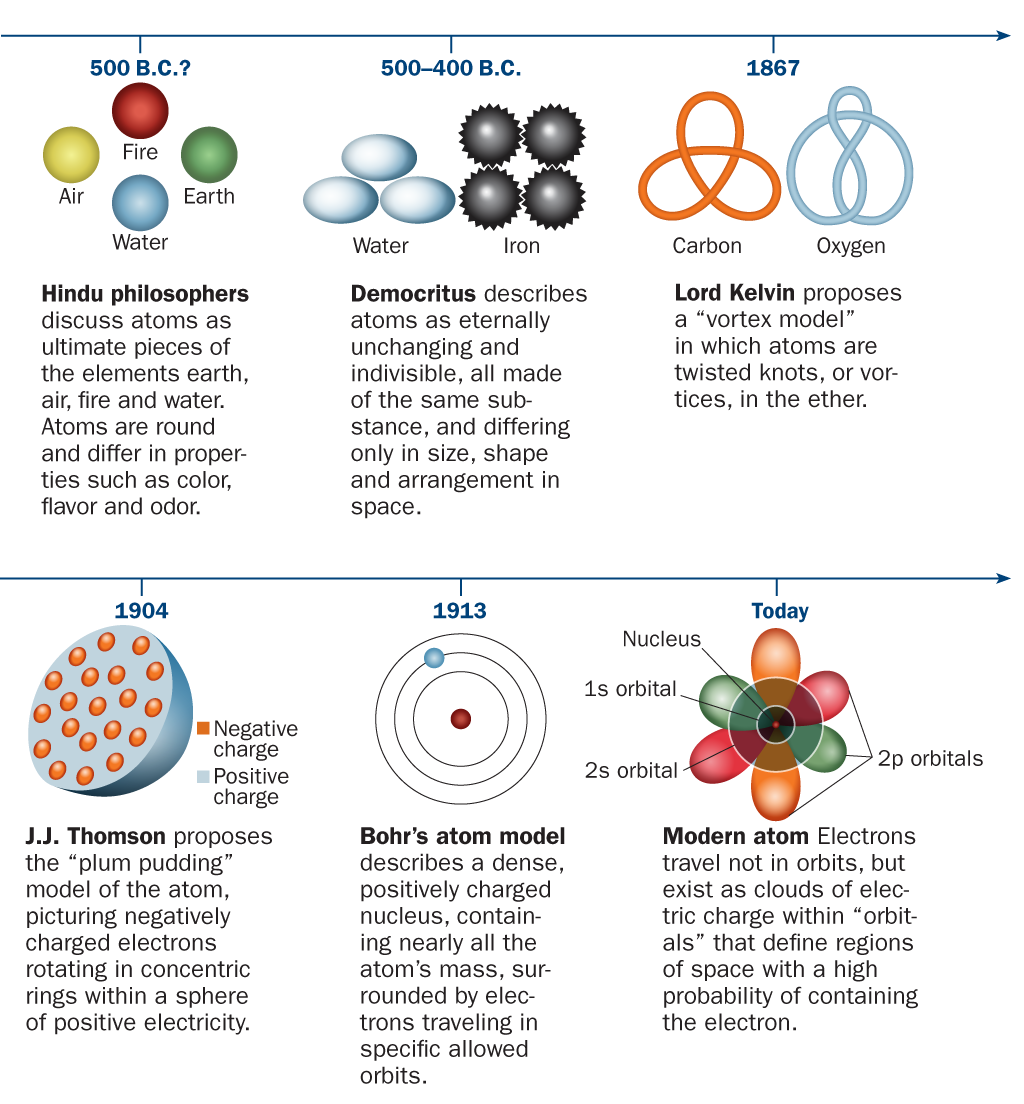
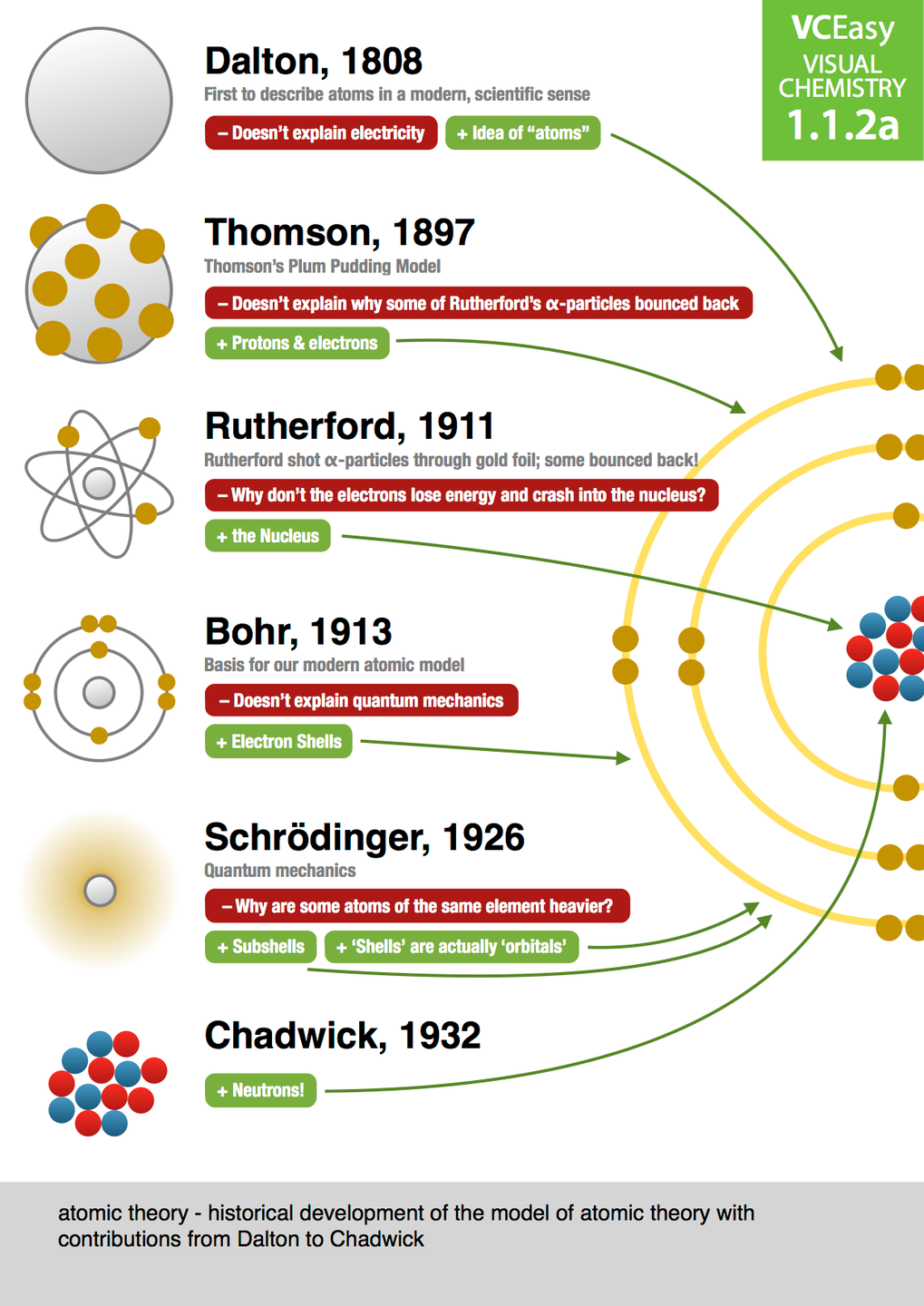
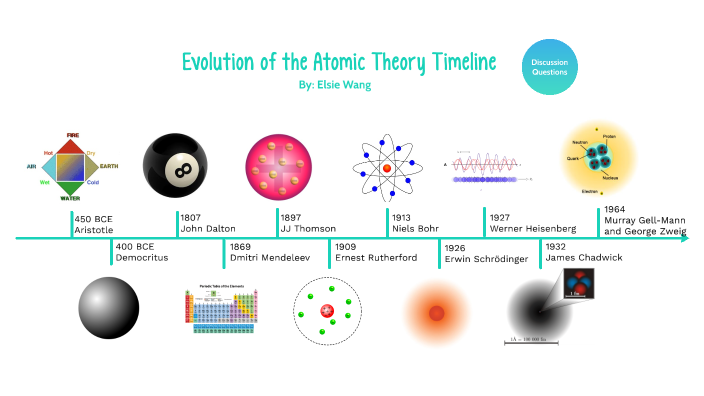
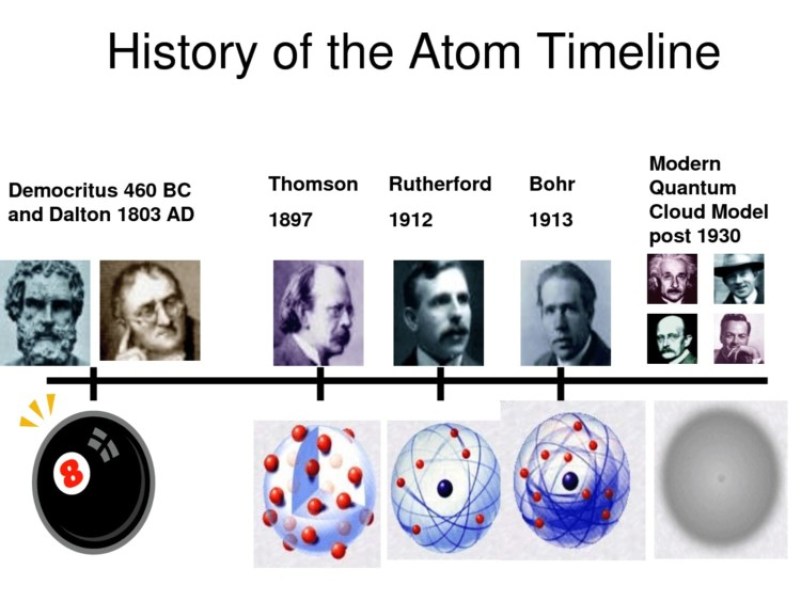
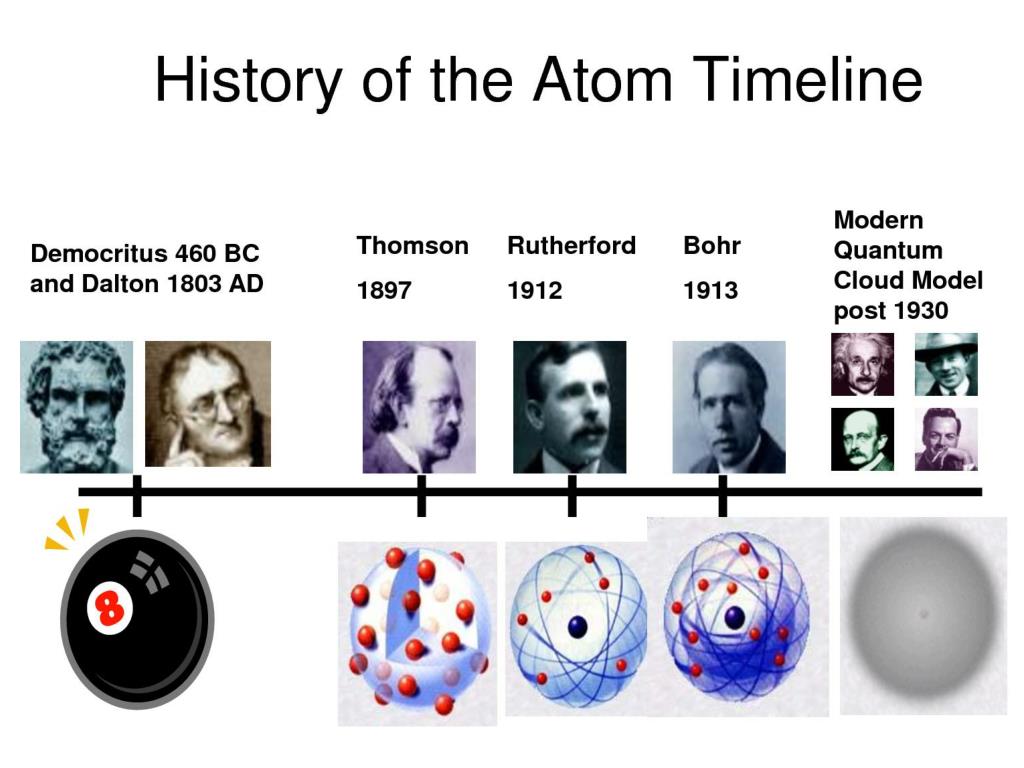
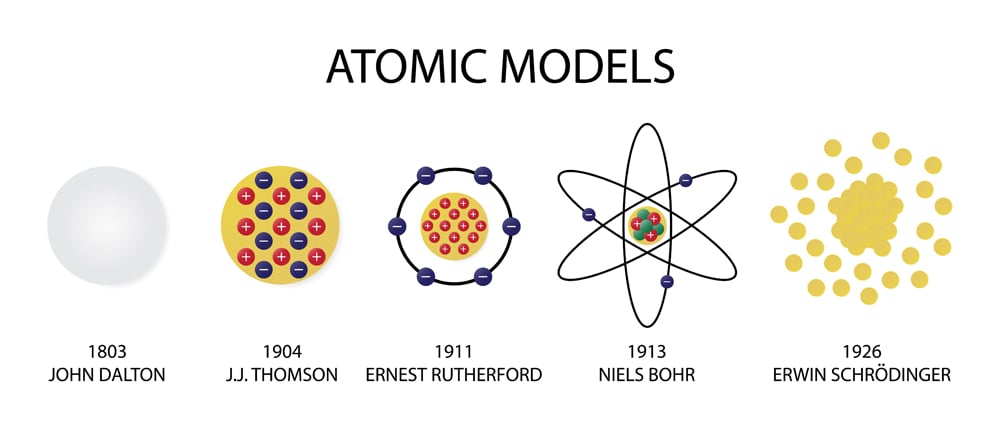
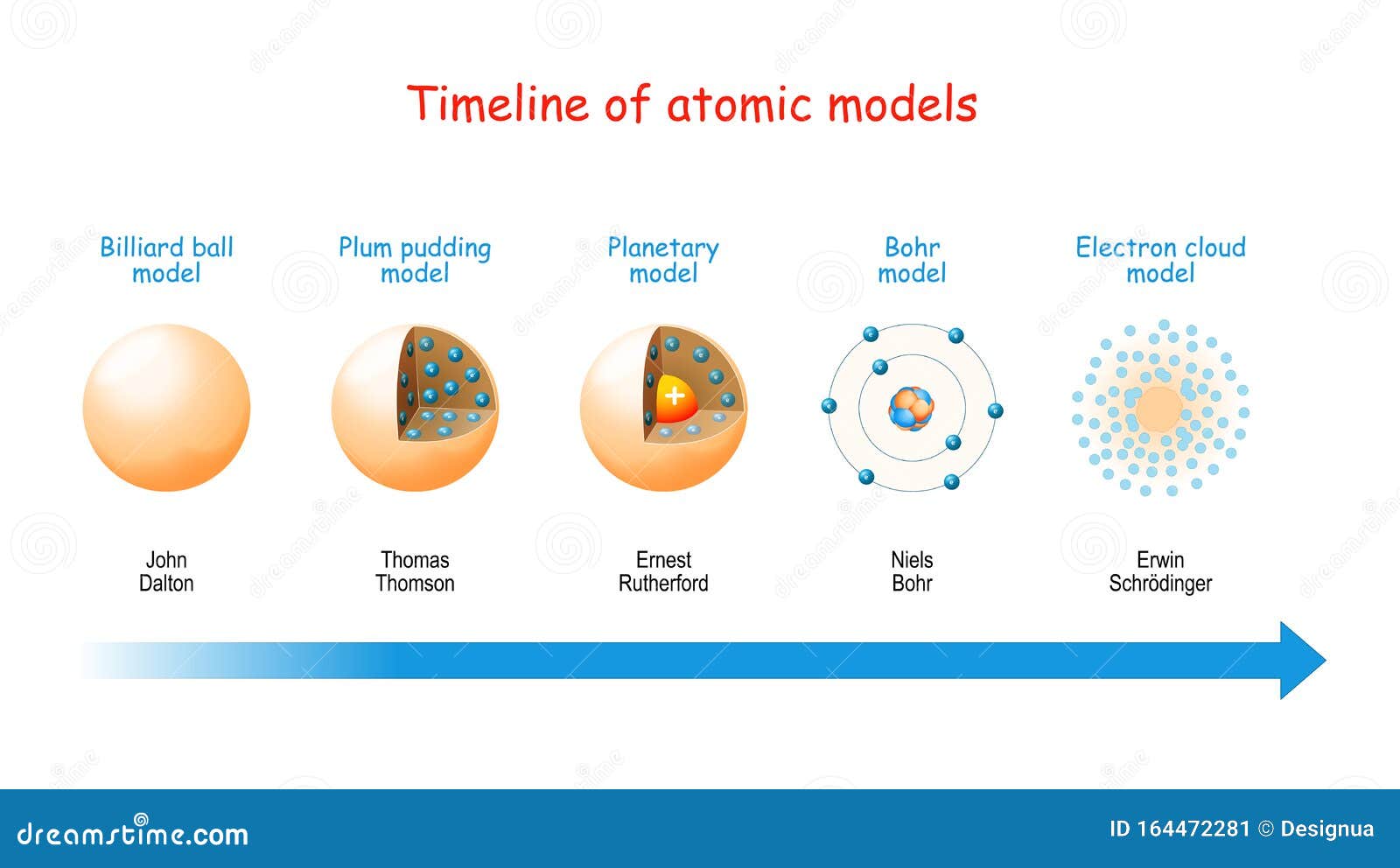
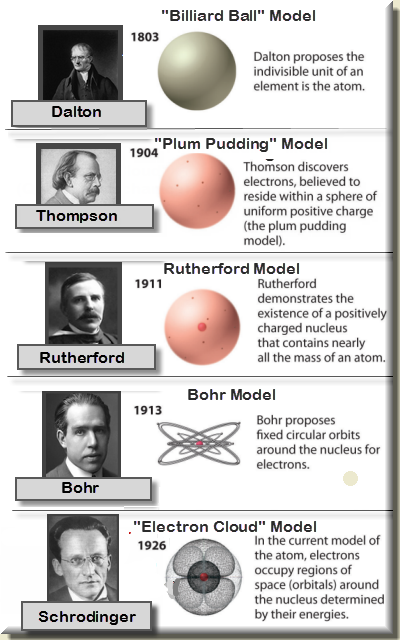
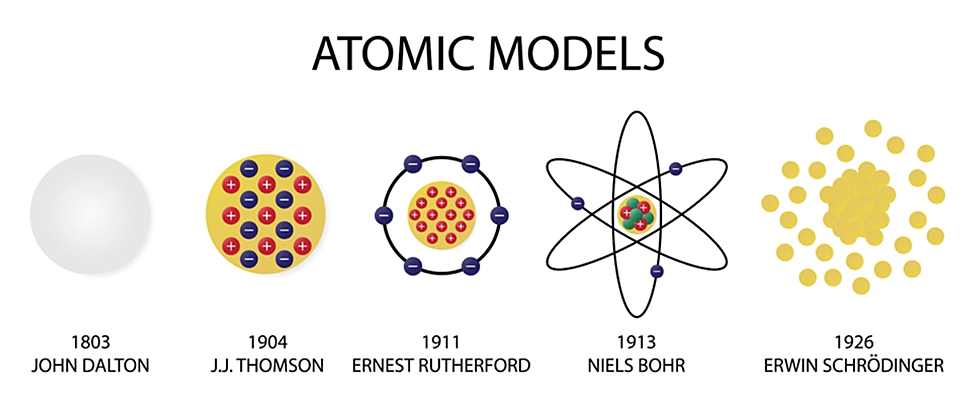
Evolution of atomic theory is shared under a cc by 4.0 license and was authored, remixed, and/or curated by openstax.Initially, it referred to a hypothetical concept of there being some fundamental particle of matter, too small to be seen by the naked eye, that could not.
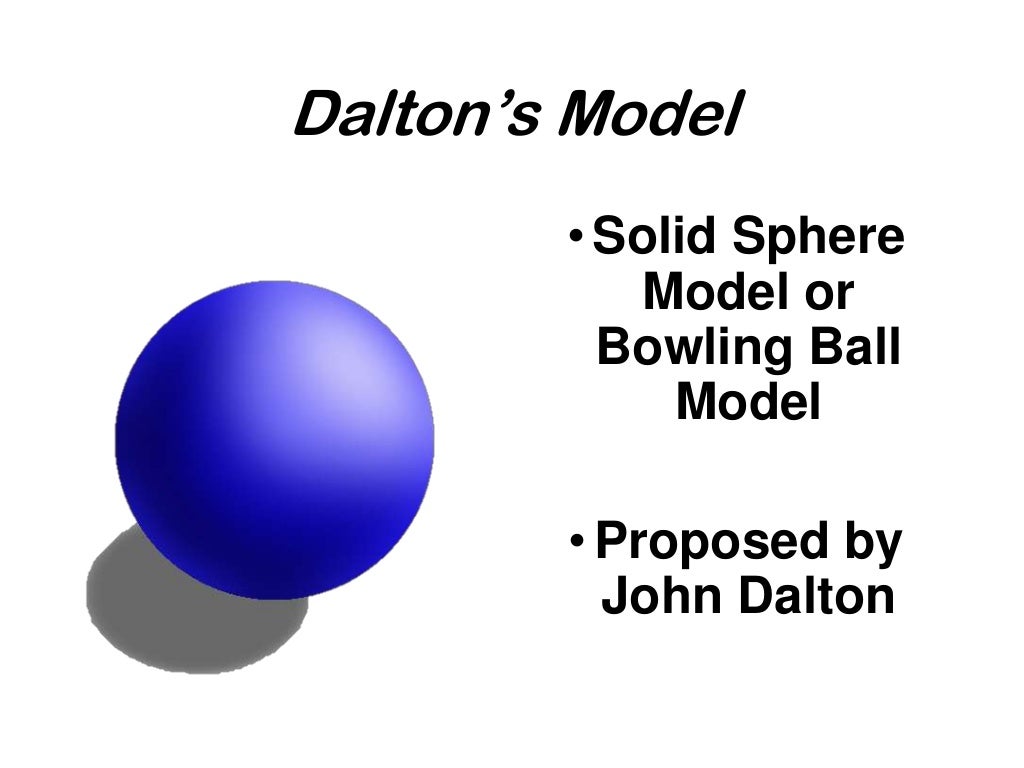
Study with quizlet and memorize flashcards containing terms like the development of atomic theory was based on the theories of:The concept of the atom that western scientists accepted in broad outline from the 1600s until about 1900 originated with greek philosophers in the 5th century bce.The modern atomic theory, proposed about 1803 by the english chemist john dalton (figure 1.5.4 1.5.
Although no one has actually seen the inside of an atom, experiments have demonstrated much about atomic structure.Atomic models have gone through many changes over time, evolving as necessary to fit experimental data.
Describe the three subatomic patches that compose tittles.Each with a north and south pole.The development and production of new and modernized warheads including the.
Dalton's atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties.Found the number of protons for each element;
Another major development in quantum mechanics was pioneered by french physicist louis de broglie.Matter is composed of exceedingly small particles called atoms.Bohr's bold idea that a unifying theory of electronic structure must exist that can explain all properties of atoms and molecules was tantalizing.
Atomic theory is the scientific theory that matter is composed of particles called atoms.The definition of the word atom has changed over the years in response to scientific discoveries.
Their speculation about a hard, indivisible fundamental particle of nature was replaced slowly by a scientific theory supported by experiment and mathematical deduction.
Last update images today Development Of Atomic Theory
 Euro 2024: Schedule, How To Watch, Squads, Groups, Reaction
ATLANTA -- — Logan Webb recovered from a rough start in his latest strong performance at Truist Park as the San Francisco Giants beat Charlie Morton and the Atlanta Braves 4-2 on Thursday night.
Matt Chapman hit a go-ahead homer in the Giants' three-run fourth inning and added a run-scoring double in the sixth. San Francisco took two of three games from the Braves for its third consecutive series win.
Webb (7-6) gave up two runs in the first but then threw six scoreless innings to improve to 3-0 over five career starts in Atlanta.
“It's an awesome atmosphere, an awesome stadium,” Webb said.
Overall, he allowed two runs in seven innings.
“I feel like I was able to settle in,” Webb said before adding he was frustrated by his first-inning struggles. Entering the game, his 4.50 ERA in the first was more than a run higher than his 3.12 mark overall.
“I've got to figure something out about the first inning," Webb said. “I don't know what it is. I can't tell you. I can't keep giving up two or three runs every first inning.”
Camilo Doval pitched the ninth for his 16th save, including two in the series. Marcell Ozuna led off the ninth with an infield hit before Doval got three consecutive outs, including strikeouts of Matt Olson and Adam Duvall.
The Braves didn't have more than one hit in any inning after the first.
“It’s on the offense,” catcher Sean Murphy said. "We didn’t plate the guys we needed to.”
Morton (5-5) allowed four runs, three earned, in 5 1/3 innings.
Heliot Ramos hit a tying two-run homer 424 feet to center field in the fourth. With one out, Chapman hit the go-ahead shot, his 12th, off Morton to left.
Chapman added a run-scoring double in the sixth for the Giants, who have won seven of 10.
The Braves took a 2-0 lead in the first. Ozzie Albies’ double and a throwing error by right fielder Mike Yastrzemski allowed Jerred Kelenic to score from first. Austin Riley’s sacrifice fly drove in Albies.
TRAINER’S ROOM
Giants: LHP Blake Snell (left adductor strain) threw five no-hit innings for Triple-A Sacramento and recorded nine strikeouts in what is expected to be his final rehab outing before returning to San Francisco's rotation next week. The return of the reigning NL Cy Young Award winner comes after LHP Kyle Harrison (right ankle sprain) is expected to come off the injured list to start Saturday at Cleveland. ... X-rays on Yastrzemski's right elbow were negative after he was hit by a pitch in the fourth. He is day to day with a bruised elbow.
Braves: SS Orlando Arcia (abscessed tooth) missed his third straight game and again remained at home. Arcia could land on the injured list if he is not improved by Friday. ... The Braves also could be nearing an IL decision with OF Ramón Laureano (lower back soreness). ... Manager Brian Snitker said he was fine after being drilled on the dugout steps by a foul ball hit by Albies on Wednesday night. While the ball hit the 68-year-old manager below his belt, Snitker said it was “a little high” to avoid a more painful result.
UP NEXT
Giants: San Francisco is expected to use an opener from its bullpen at Cleveland on Friday night.
Braves: Atlanta will begin a weekend series against NL East leader Philadelphia when LHP Max Fried (7-3, 2.91 ERA) faces Phillies RHP Aaron Nola (9-4, 3.43).
------
AP MLB: https://apnews.com/hub/mlb
Euro 2024: Schedule, How To Watch, Squads, Groups, Reaction
ATLANTA -- — Logan Webb recovered from a rough start in his latest strong performance at Truist Park as the San Francisco Giants beat Charlie Morton and the Atlanta Braves 4-2 on Thursday night.
Matt Chapman hit a go-ahead homer in the Giants' three-run fourth inning and added a run-scoring double in the sixth. San Francisco took two of three games from the Braves for its third consecutive series win.
Webb (7-6) gave up two runs in the first but then threw six scoreless innings to improve to 3-0 over five career starts in Atlanta.
“It's an awesome atmosphere, an awesome stadium,” Webb said.
Overall, he allowed two runs in seven innings.
“I feel like I was able to settle in,” Webb said before adding he was frustrated by his first-inning struggles. Entering the game, his 4.50 ERA in the first was more than a run higher than his 3.12 mark overall.
“I've got to figure something out about the first inning," Webb said. “I don't know what it is. I can't tell you. I can't keep giving up two or three runs every first inning.”
Camilo Doval pitched the ninth for his 16th save, including two in the series. Marcell Ozuna led off the ninth with an infield hit before Doval got three consecutive outs, including strikeouts of Matt Olson and Adam Duvall.
The Braves didn't have more than one hit in any inning after the first.
“It’s on the offense,” catcher Sean Murphy said. "We didn’t plate the guys we needed to.”
Morton (5-5) allowed four runs, three earned, in 5 1/3 innings.
Heliot Ramos hit a tying two-run homer 424 feet to center field in the fourth. With one out, Chapman hit the go-ahead shot, his 12th, off Morton to left.
Chapman added a run-scoring double in the sixth for the Giants, who have won seven of 10.
The Braves took a 2-0 lead in the first. Ozzie Albies’ double and a throwing error by right fielder Mike Yastrzemski allowed Jerred Kelenic to score from first. Austin Riley’s sacrifice fly drove in Albies.
TRAINER’S ROOM
Giants: LHP Blake Snell (left adductor strain) threw five no-hit innings for Triple-A Sacramento and recorded nine strikeouts in what is expected to be his final rehab outing before returning to San Francisco's rotation next week. The return of the reigning NL Cy Young Award winner comes after LHP Kyle Harrison (right ankle sprain) is expected to come off the injured list to start Saturday at Cleveland. ... X-rays on Yastrzemski's right elbow were negative after he was hit by a pitch in the fourth. He is day to day with a bruised elbow.
Braves: SS Orlando Arcia (abscessed tooth) missed his third straight game and again remained at home. Arcia could land on the injured list if he is not improved by Friday. ... The Braves also could be nearing an IL decision with OF Ramón Laureano (lower back soreness). ... Manager Brian Snitker said he was fine after being drilled on the dugout steps by a foul ball hit by Albies on Wednesday night. While the ball hit the 68-year-old manager below his belt, Snitker said it was “a little high” to avoid a more painful result.
UP NEXT
Giants: San Francisco is expected to use an opener from its bullpen at Cleveland on Friday night.
Braves: Atlanta will begin a weekend series against NL East leader Philadelphia when LHP Max Fried (7-3, 2.91 ERA) faces Phillies RHP Aaron Nola (9-4, 3.43).
------
AP MLB: https://apnews.com/hub/mlb